#acoziborole-drug-approval
#acoziborole-drug-approval
[ follow ]
#alzheimers-disease #clinical-trials #glp-1-drugs #addiction-treatment #gene-therapy #dravet-syndrome #glp-1
fromenglish.elpais.com
1 day agoFollowing the initial trials in Africa of the groundbreaking drug that could put an end to AIDS
On that sunny March morning, in a small health center in Lobamba, a rural area of Eswatini, this 32-year-old sex worker has just become one of the first people in the world to receive lenacapavir, a drug that, administered twice a year, offers nearly 100% protection against HIV.
Medicine
Artificial intelligence
fromTNW | Startups-Technology
2 weeks agoRivia raises 13M to bring agentic AI to clinical trials
Rivia raised €13 million to build AI agents that actively manage clinical trial operations by surfacing insights and flagging anomalies, advancing beyond traditional data storage systems.
fromABC7 New York
4 days agoFDA grants speedy approval to Eli Lilly's weight-loss pill for obesity
The Food and Drug Administration granted expedited approval to orforglipron, a GLP-1 drug that works like widely used injectable medications to mimic a natural hormone that controls appetite and feelings of fullness.
Medicine
from24/7 Wall St.
3 weeks agouniQure, Syndax and Erasca Are Drawing Analyst Interest Ahead of Key Drug Catalysts
RBC Capital analyst Luca Issi upgraded the stock to Outperform from Sector Perform with a price target of $35, up from $11. Wells Fargo also upgraded uniQure to Overweight from Equal Weight with a $60 price target. The catalyst: the departure of Vinay Prasad from the FDA. RBC views this as a positive for uniQure, noting it is "not inconceivable" that the FDA reverts to its prior stance, and believes Prasad's departure is likely to open up a more balanced discussion on risk/reward for Huntington's disease.
NYC startup
Healthcare
fromFast Company
1 month agoResponsible compounding could close the innovation gap
Compounding can responsibly accelerate patient access to needed therapies when grounded in rigorous data, filling genuine clinical gaps while pursuing FDA approval, particularly in underserved areas like women's health.
Medicine
fromNews Center
2 weeks agoSchizophrenia Study Finds New Biomarker, Drug Candidate to Treat Cognitive Symptoms - News Center
Northwestern researchers identified a novel schizophrenia biomarker in cerebrospinal fluid that could enable new treatments for cognitive symptoms through a synthetic protein therapeutic approach.
#gene-therapy
Medicine
fromNews Center
3 weeks agoFirst Gene Regulation Clinical Trials for Epilepsy Show Promising Results - News Center
Zorevunersen, a gene-regulation therapy, demonstrates safety and effectiveness in reducing seizures and improving developmental outcomes in Dravet syndrome patients by targeting the underlying genetic cause.
fromwww.theguardian.com
1 month agoPsychiatric drugs aren't always the answer | Letter
Yes, there has been a shocking lack of progress in developing transformative psychiatric medicine (We need new drugs for mental ill-health, 5 February), but this may be because in mental health, drugs are not always the answer (see, for example, Richard P Bentall's Doctoring the Mind). Huge progress has been made in the effectiveness of talking therapies for example, free effective treatment for post-traumatic stress disorder (PTSD) is available to all UK army veterans through the charity PTSD Resolution.
Mental health
Science
fromNews Center
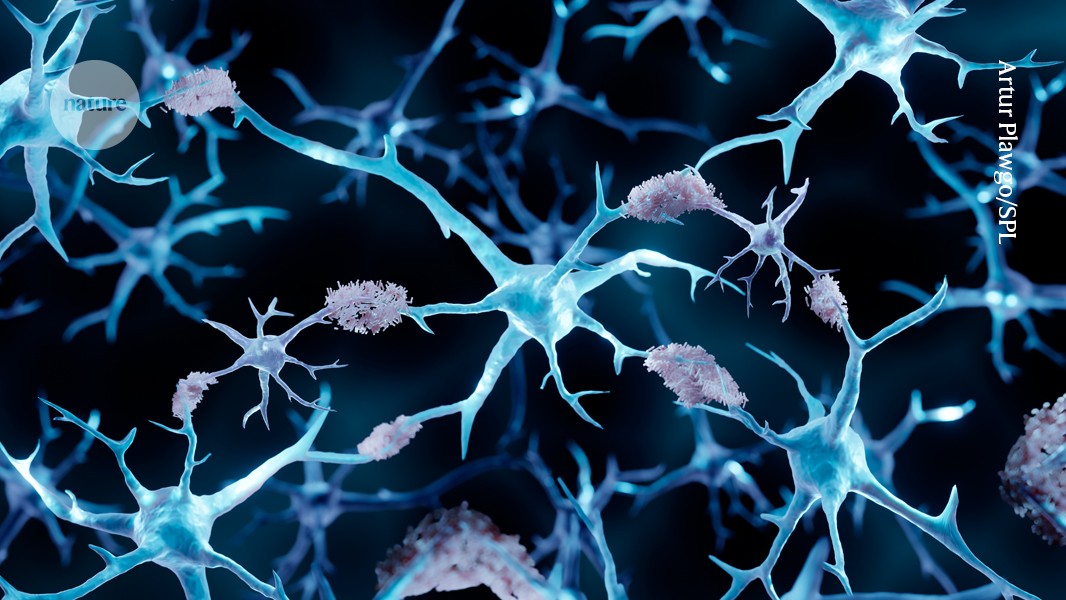
2 months agoTargeting Key Proteins in Fight Against ALS - News Center
RAD23 controls both degradation and stabilization of misfolded proteins; reducing RAD23 enhances clearance of disease-linked aggregates, offering a therapeutic target for neurodegenerative proteostasis dysfunction.
Medicine
fromwww.theguardian.com
3 weeks agoRecreational drugs can more than double risk of stroke, study suggests
Amphetamine and cocaine use more than double stroke risk, with cannabis increasing risk by 37%, while opioids show no increased stroke risk according to analysis of over 100 million people.
fromSilicon Canals
1 month agoA brain-based AI test could point to the best antidepressant for you - Silicon Canals
Before treatment began, participants underwent neuroimaging. Instead of relying on a single modality, the researchers fused structural connectivity (how regions are physically wired) with functional connectivity (how regions co-activate at rest). The goal was not to throw every possible feature at a black box, but to learn a constrained pattern-what the authors call structure-function "covariation"-that carries the most predictive signal for outcome. In other words, the model tries to find the smallest set of connections that meaningfully forecasts symptom change.
Mental health
fromPsychology Today
1 month agoZuranolone: A Novel Treatment for Postpartum Depression
For most cases of mild to moderate postpartum depression, standard treatment with psychotherapy with or without an antidepressant works well. Treating severe PPD is more challenging. Fortunately, there is a new category of medication for PPD that is intended for severe PPD. The newest one is zuranolone (trade name is Zurzuvae). Currently, "existing efficacy data are limited to individuals with severe postpartum depression, where long-term outcomes need further study" (Miller, 2025). It has a different mode of action than older antidepressants.
Mental health
fromPsychology Today
1 month agoLow Dose Sublingual Ketamine
Statistics show that about one-third of people with depression achieve remission-meaning their symptoms are gone-with traditional antidepressant medications. This matched my experience treating people, and I had grown to accept that this was as good as it gets. Although I wasn't thrilled with the fact that many people continued to struggle with significant symptoms of persistent depression, it seemed this was as good as we could do.
Mental health
fromwww.scientificamerican.com
1 month agoAlzheimer's blood tests may predict when a person will develop symptoms
But questions remain about the accuracy and uncertainty of these tests, and experts caution that the assays aren't ready for prime time. While the results here are encouraging, they are not yet at the level of having significant clinical benefit for individual patients, says Corey Bolton, a clinical neuropsychologist and an assistant professor of medicine at Vanderbilt University Medical Center, who was not involved in the new study.
Medicine
fromwww.theguardian.com
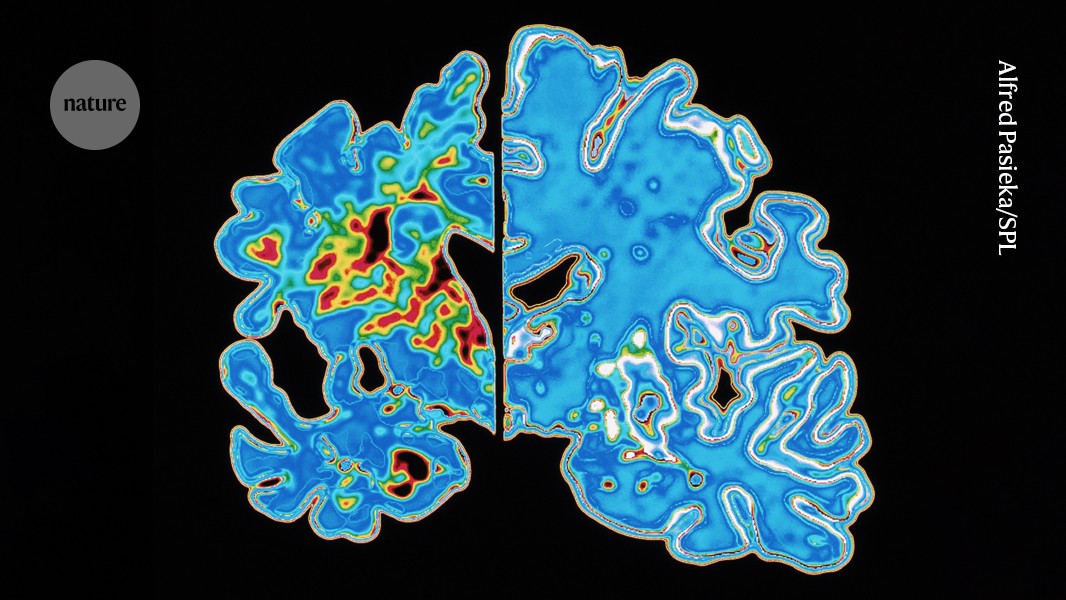
2 months agoAlzheimer's therapies should target a particular gene, researchers say
New therapies for Alzheimer's disease should target a particular gene linked to the condition, according to researchers who said most cases would never arise if its harmful effects were neutralised. The call to action follows the arrival of the first wave of drugs that aim to treat Alzheimer's patients by removing toxic proteins from the brain. While the drugs slow the disease down, the benefits are minor,
Medicine
fromTNW | Deep-Tech
1 month agoAerska raises $39M to help RNA medicines reach the brain
For families living with neurodegenerative disease, the hardest part is not always the diagnosis. It is the slow erosion that follows: memory fading, personality shifting, independence shrinking. It unfolds quietly. First, forgotten appointments. Then repeated questions. Then moments when a familiar face no longer feels familiar. The illness does not isolate itself to one body. It rearranges the lives around it.
Medicine
from24/7 Wall St.
2 months agoIs Apellis Pharmaceuticals' FDA Win Just the Beginning?
EMPAVELI is the first and only approved treatment for C3G and IC-MPGN across pediatric patients 12+, adults, and post-transplant recurrence. That's roughly 5,000 patients in the U.S., with EMPAVELI holding exclusive approval for about two-thirds. Add the European CHMP positive opinion in December 2025, and you have a rare disease franchise with global expansion potential and pricing power that typically commands gross margins north of 90%.
Medicine
fromNews Center
2 months agoExperimental Drug Shows Promise for Rare Genetic Disorder - News Center
Mucopolysaccharidosis type II (MPS II), or Hunter syndrome, is a rare genetic disorder primarily affecting boys, caused by a deficiency in the enzyme needed to break down sugar molecules. This harmful buildup in cells and tissues impacts multiple body systems, causing frequent infections, organ enlargement and developmental disabilities. Management involves supportive care and enzyme replacement therapy, as there is currently no cure,
Medicine
fromnews.feinberg.northwestern.edu
2 months agoNew Institute Envisions Future Where Our Brains Last as Long as Our Bodies - News Center
Northwestern University has launched the Simpson Querrey Brain Health Institute (SQ-Brain), made possible by nearly $25 million in philanthropic funding from university trustee Kimberly K. Querrey ('22, '23 P). SQ-Brain envisions a future where our brains last as long as our bodies a world where brain health is continuously measurable, modifiable and monitorable across the lifespan, and where prevention of cognitive decline and brain injury is anchored in neurovascular biology and precision medicine.
Medicine
fromCbsnews
2 months agoNovo Nordisk debuts Wegovy weight-loss pill in the U.S.
A pill form of weight-loss drug Wegovy, manufactured by Danish drugmaker Novo Nordisk, is now available in the U.S., giving Americans access to an oral medication to treat obesity. The starting dose of the daily Wegovy pill, at 1.5 milligrams, is now available for $149 per month for self-paying patients, Novo Nordisk announced on Monday. A 4 mg dose of the medication is available for the same price through April 15, after which the cost will rise to $199.
Medicine
fromFast Company
2 months agoFDA commissioner's drug review plan sparks alarm across the agency
The Food and Drug Administration commissioner's effort to drastically shorten the review of drugs favored by President Donald Trump's administration is causing alarm across the agency, stoking worries that the plan may run afoul of legal, ethical, and scientific standards long used to vet the safety and effectiveness of new medicines. Marty Makary's program is causing new anxiety and confusion among staff already rocked by layoffs, buyouts, and leadership upheavals, according to seven current or recently departed staffers.
Medicine
Medicine
fromTechCrunch
2 months agoFrom OpenAI's offices to a deal with Eli Lilly - how Chai Discovery became one of the flashiest names in AI drug development | TechCrunch
AI-driven startups like Chai Discovery accelerate antibody design, attracting major funding and partnerships with pharma and tech companies to transform drug discovery.
fromNews Center
2 months agoPost-Stroke Injection Protects the Brain in Preclinical Study - News Center
When a person suffers a stroke, physicians must restore blood flow to the brain as quickly as possible to save their life. But, ironically, that life-saving rush of blood can also trigger a second wave of damage - killing brain cells, fueling inflammation and increasing the odds of long-term disability. Now, in a study published in the journal Neurotherapeutics, Northwestern University scientists have developed an injectable regenerative nanomaterial that helps protect the brain during this vulnerable window.
Medicine
[ Load more ]